1. First you must understand how DC current (direct current) works
DC CURRENT: ALL Footbath electrolysis units on the market are using DC current. They either run from an AC input or a battery input (a battery is DC). This is necessary to create the electrolysis process. Direct current or DC electricity is the continuous movement of electrons from negative to positive through a conducting material such as a metal wire or in the case of the footbath it is the water. A DC circuit is necessary to allow the current or steam of electrons to flow. The current ALWAYS flows from the negative (Cathode ring) to the positive (Anode ring) through the water. When the electrons move from the Cathode ring to the Anode ring it causes the Cathode ring to deplete and then binds with molecules in the water and then moves to the Anode ring creating a plating on the Anode ring (refer to our research page showing the scrapings of the Anode ring click here). When a machine switches polarity ALL THEY ARE DOING is changing which plates receive the positive and the negative. The current STILL flows from Negative to Positive. Remember the Cathode ring depletes the metal & the Anode ring attracts where a build up occurs, so when the Anode ring switches to receiving the negative, making it now the Cathode ring, it then releases all the build-up back in to the water. Not only are you releasing the toxins back in to the water but it gains NO BENEFIT. (click here for outside source)
False misleading statement by other companies state (i.e. AMD IONCLEANSE) when the unit is supposedly in ‘positive’ mode: “When the unit is in positive mode, the current flows to the positive pole in the array, which results in more negative ions being produced in the water. When it is set on negative mode, the current flows to the negative pole in the array, which results in more positive ions being produced in the water. Therefore, negative polarity generates a positive ion flow. This process is known as the electrolysis of water.” stated by AMD Ioncleanse. ** THIS IS FALSE: A DC current cannot move from positive to negative pole as stated in the negative mode. (explained above)
A VIDEO from AMD IONCLEANSE that demonstrates ‘INCORRECT’ info on DC Current
https://www.youtube.com/watch?v=MpuKOtFBztU
2. Second you need to understand how Water Electrolysis works?
WATER ELECTROLYSIS is the process by which water is broken down into hydrogen and oxygen in the presence of a direct electric current and an electrolyte (NACL salt). Electrolysis of water is what’s called a redox reaction. This means that electrons are being moved from one molecule to another. To electrolyze water, you need 2 electrodes, the cathode (negative) and the anode (positive). This would be the water module of the footbath unit. You also need to dissolve ions in the water to improve its conductivity; sodium chloride (salt) is often used as in the case of the footbath.
At the anode, electrons are removed from the water and it is oxidized, in this reaction:
2H2O —–> O2 + 4H+ + 4e–
At the cathode, electrons are added to water and it is reduced:
2H2O + 4e– —–> 2H2 + 4OH–
So at the anode we get oxygen gas and hydrogen ions, at the cathode hydrogen gas and hydroxide ions.
Other products can be formed depending on what the electrodes are made from and how the electrolysis chamber is built. If the cathode is made from iron, it will react with the hydroxide ions to make brown, insoluble iron hydroxide:
Fe3+ + 3OH– —–> Fe(OH)3
Chlorine gas can be formed, but this isn’t because the electrodes are made of carbon. It will only happen if sodium chloride is added to the water. In this case, the chloride ions in the water can oxidise to chlorine and combine with hydroxide ions to form hypochlorite:
Cl2 + 2OH– —–> Cl– + OCl– + H2O
False misleading statement by other companies that use the DUAL Polarity claim that their machines can create more positive ions or negative ions depending on what mode the machine is in (positive or negative). This is a false statement. The current coming from the water module acts as a stimulus or Optimizer to break up the H2O (water) and the NACL (salt). Current will always flow the same way, meaning only from negative to positive (as stated above), so the breakup of the water molecule & salt will be exactly the same. The molecule makeup of the H2O and the NACL is what determines how many negative ions and positive ions are being produced NOT THE CURRENT (measured in e-). Just by adding salt this creates about a 2 to 1 ratio of negative ions (just like the ocean).
An example of this false statement is stated on AMD IONCLEANSE webpage. They state that depending on what ‘supposed’ mode (positive/negative) their unit is set to, affects differently the combination of Hydrogen & Oxygen gas release. They say it breaks down in:
FALSE:
POSITIVE MODE: 4H2O + 4e- à 2H2 + 4OH- OR
NEGATIVE MODE: 2H2O à O2 + 4H+ + 4e-.
This is a false statement because as you can see above where we explain how ‘water electrolysis‘ works, the electrolysis creates at the anode water is oxidized (2H2O —–> O2 + 4H+ + 4e–) and at the cathode water is reduced: (2H2O + 4e– —–> 2H2 + 4OH– )
END RESULT OF ALL FOOTBATH ELECTROLYSIS PROCESS: The overall effect from the footbath electrolysis will always breakdown in to 2H_2 0 = 2H_2 + O_2 (details click here from American Chemical Society
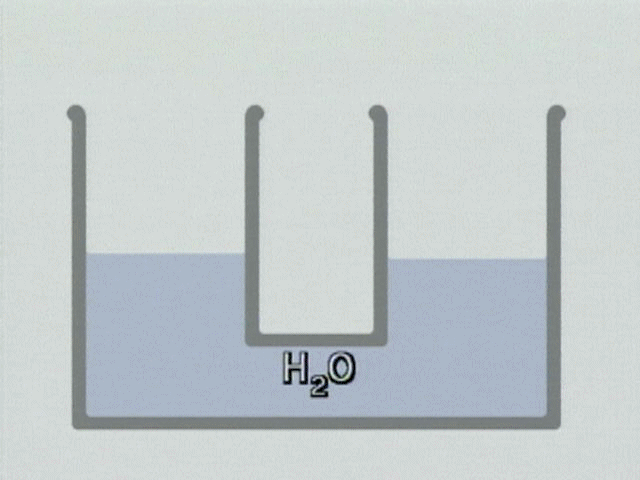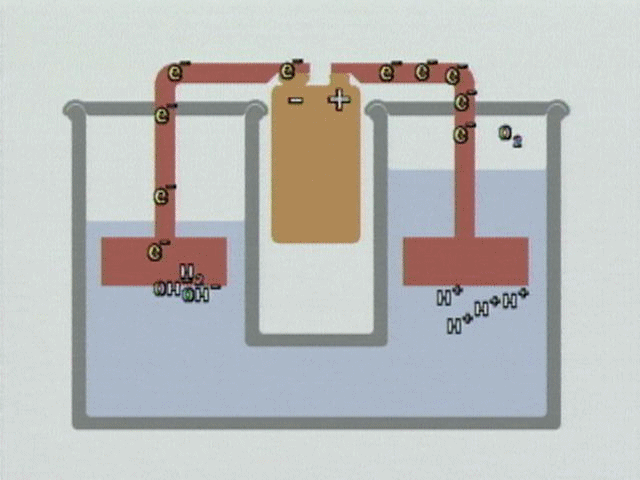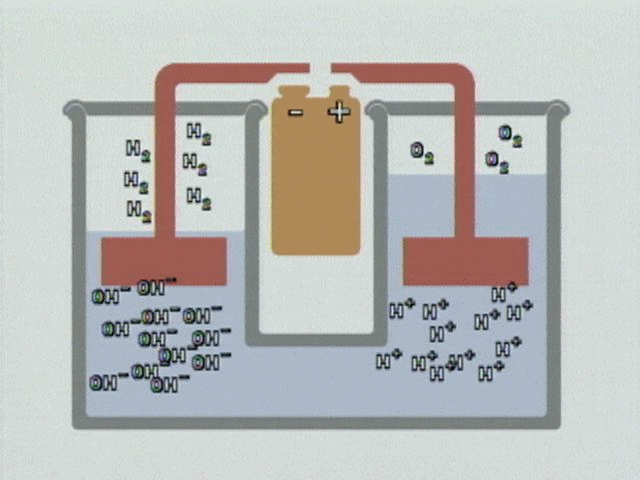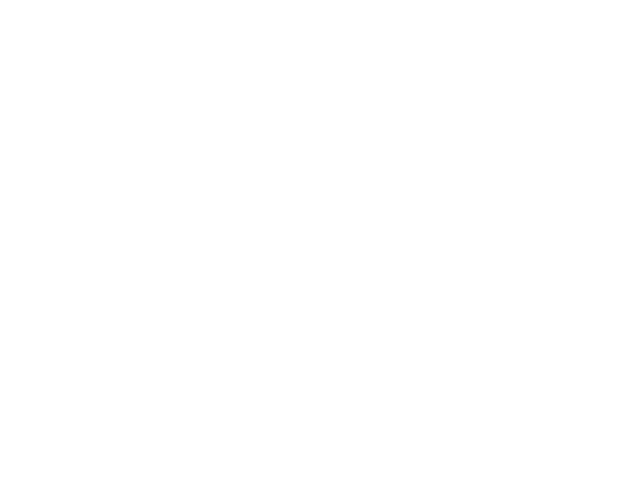
For full video demo. https://www.chemedx.org/video/electrolysis-neutral-solution-animation
